Exquisite equations
- Jan 12, 2025
- 7 min read
In the summer of 2022, I got a bit bored, and may have spent an hour compiling my favourite equations and sketches of my degree into a funky wallpaper on my tablet, all by hand. I have since then covered another two more years of physics, so potentially my wallpaper could do with an update. All of these equations are ones I have used at various points, and some may be recognisable to a non-physicist too. They range from trigonometric functions used in A level Maths to tiny scale quantum mechanics and large scale cosmology. One day, I may become a physics or maths teacher, and I would particularly like to be the headmistress of a girls' school like the one I have happy memories of, so think of this as a little taster for any future pupils to whom I may impart some 'pearls of wisdom'. (This was a genuine quote from one of my favourite maths teachers, Mr Rouse, during a lesson on integration by parts, or possibly mechanics, either way he taught us a lot about both).
I'll pick out my favourite equations below.

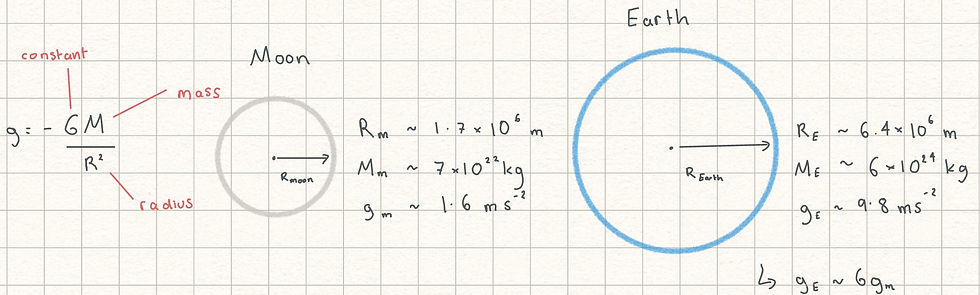
Gravity - and ball games on the moon
An extremely important phenomena without which life as we know it would not be possible: galaxies, stars and planets wouldn't form, and we would have nothing keeping our feet on the ground, to state just a few headaches which may arise in a gravity-less universe. Gravity is the force per unit mass an object experiences, which can also be thought of as an acceleration. So that this post doesn't turn too much into an abstract physics lesson, think of two experiments. In the first, you're standing on Earth in a park on a normal day, and you kick a ball upwards. Depending how hard you kick it, it'll go up quickly, then slow down to a stop at its highest point, before coming back down again, accelerating as it does so. The exact value of this acceleration is 9.81 ms^-2, which means the speed increases by 9.81 metres per second, for every second that it falls. This is why it is never a good idea to throw even small things out of a multistorey building if people are walking below, even a teacup chucked out of a window could be lethal if it falls from a few metres up. Now consider the scene in the first Wallace and Gromit film, A Grand Day Out, where our favourite plasticine characters go to the moon in a self made rocket in search for cheese on a bank holiday. Leaving aside a few scientific inaccuracies, one thing that is broadly correct is when Wallace kicks a ball on the moon, yet to his surprise it doesn't fall back down. This is because the pull of gravity is much weaker on the moon than on Earth, because the mass of the moon is much smaller than the mass of Earth (about 100 times lighter). Gravity on the moon is a mere 1.62 ms^-2, about a sixth of that on Earth. In reality what this means is if you were to jump around on the moon, you'd jump much higher than you could on Earth because the pull back down to the surface is 6x weaker. Similarly, Wallace's football can now travel much higher on being kicked before coming back down than it would on Earth. Having no air resistance on the moon due to lack of atmosphere also helps the ball travel further.
Fun fact: in 1971 with the Apollo 14 mission, NASA astronaut Alan Shepard smuggled a modified golf club and balls into his spacesuit and socks, and hit a golf ball on the moon. Unlike Wallace, he was wearing a spacesuit and his movement was therefore quite restricted, not ideal for doing sport. Nevertheless, science comes alive with mad stories like that, and now you've got images of people messing around with balls on the moon in your mind, hopefully the elusive concept of gravity is a little more interesting now.

Schrödinger's equation
The classic equation that gets thrown around on any TV show featuring a physicist and plastered on nerdy t-shirts. But what does it actually mean? A big gripe of mine is physicists overcomplicating things to make themselves look clever and other people who don't understand feel stupid, which puts people off what is actually a jolly interesting subject. This equation helps physicists predict how an electron trapped in an atom will behave. Energy comes in different forms: light, sound, heat etc. It is always conserved in closed systems, and can be neither created nor destroyed, only transferred from one form to another. In small scale physics such as quantum mechanics, the relevant types are kinetic energy (movement), and potential energy (related to charges of a nucleus, as it takes energy to escape the pull of a charged particle, in a similar way to requiring energy to kick a ball away from Earth being dragged down by gravity).

This complicated looking equation can be broken down into fairly simple components, and shows the conservation of energy for wave functions. Quantum mechanics is... a bit weird, but basically it boils down to particles are represented as waves, and described by wave functions, for some reason given the symbol Ψ, a Greek letter called Phi. Ψ depends on position and time, and from doing various operations with it one can find out a particle's momentum or position, but only probably, not definitely (yeah, weird, also it turns out you can't know both things at once or predict them, only average out an expected value over several measurements). This form of the equation above is time-independent (doesn't depend on time), so is static, and often called the 'particle in a box' example, where only the position parts of the Schrodinger equation remain, not the time ones, which makes it slightly prettier to look at on paper and easier to work with- basically, it's just a conservation of energy equation for particles.
Electron orbits
Provided one wasn't peacefully snoring (or daydreaming) through school chemistry lessons, one may faintly recall that electrons orbit nuclei. What the teachers left out at the time, was that these orbits aren't perfectly circular. Electron orbits can oscillate, depending on their energy state, and make pretty patterns such as the ones below. The oscillations must join up to make a full circle, meaning that only an integer (whole) number of waves can form in any one orbit. If you're still not sure what I'm on about, this picture below should help. In a), 4 waves fit around the circle, and in b) there are now 5 waves, indicating a high energy level. In c) however, the author has tried to draw a non-integer number of waves, which means the electron's orbit is discontinuous and not possible. This may seem a bit 'so what?', but this is the reason electrons have specific energy levels, which has implications for the composition of all matter as we know it, because these energy levels allow electrons to orbit nuclei in a stable manner, rather than spiralling inwards haphazardly, causing the atom to collapse. Bottom line, we wouldn't be here if it wasn't for electrons orbiting atomic nuclei in tidy patterns of integer numbers of waves.

Jeans mass
This is the maximum mass a cloud of gas can have (for a given temperature and density) before it collapses under its own gravity, an equation that is crucial to explaining star formation.
The drawing below illustrates roughly what happens during star formation, from the molecules in the interstellar medium, to molecular clouds, to young stars forming. The interstellar medium from which stars form is uneven in density, and if a region of it is perturbed for example by a supernova shockwave passing through, part of the molecular cloud can be compressed. A clump of mass will have its own gravity, which gets stronger as the clump get more massive. Eventually, it collapses under its own gravity, and this is what kickstarts heating and star formation.

With reference to the Jeans mass now: it is important to note this is not a constant, as it varies depending on the cloud's temperature and density. The most common stars in the universe by a long way are low mass stars, as somehow the conditions of molecular clouds form these preferentially over high mass (therefore brighter) stars. Nevertheless, there are massive stars around, so somehow it is possible to form them, and the mechanisms behind this still puzzle astronomers. One issue relating to the Jeans mass is that clouds tend to fragment (for reasons of energy balance which can be read about in my notes below), and fragmented clouds are smaller and therefore have less material from which stars can grow, making them less massive.

For the mathematically minded, find below my revision notes from last year's exam on this very topic*. That first class physics degree didn't just earn itself, turns out some students really do get studying done at university... ;)
Square roots
A nice easy one to finish with which readers may remember from GCSE maths, this equation will find the real roots of a quadratic equation, and may help with any graph drawing subsequently required (a niche yet satisfying skill). An example is shown below.

If no real roots can be found, then we get into the realm of imaginary numbers - they sound made up but they're actually a very interesting branch of mathematics with some useful applications to the, ah, real world.
So there we have it, some random equations which are hopefully not quite as random and intimidating now, and actually quite cool. Turns out you can describe physical phenomena in equations and mathematics as well as in words. Does that make physicists automatically billigual?
*Derivation of the Jeans mass
With thanks to Pablo for the lovely Stars from Birth to Death module.





Comments